Water in the Atmosphere
Although I previously wrote on the topic of “Water,” here I would like to narrow my focus to specifically on “Water in the Atmosphere.” I am doing this for three reasons. First, it provides a great example from our immediate surroundings where one can explain basic scientific principles in a simple manner to non-scientists. Second, the topic affects us all on a daily basis whether we’ve ever thought about it or not. How can I dry the clothes faster? Why do I sweat? How can I defog the windshield? And where does rain come from anyway? Third, an understanding of the concepts presented here provides a foundation for future posts I’d like to write on hurricanes, the greenhouse effect and global climate change.
Water and Air Together
It is possible that you have heard the concept that “warm air holds more water than cold air.” This is a somewhat misleading and mysterious statement which will become more clear once we actually see what happens when water and air come together. So, we will start by imagining a simple scenario: a closed jar, half filled with water and with perfectly dry air above it at room temperature and sea level pressure (the weight of all the air above you as you stand next to the ocean). I touched on the concept of temperature in my essay on “Fire“, but here I will explain it more carefully. All atoms and molecules around us are jiggling more quickly or more sluggishly depending on how much kinetic energy they each have. We do not feel these individual molecules though of course. What we experience as temperature, whether we measure it through a thermometer or our fingertips, is an average of the jiggling of the molecules. In the diagram below we can compare curves of how many molecules are jiggling at each kinetic energy level at two different temperatures.

This is a graph showing the distribution of molecular kinetic energy level at two different temperatures. The left (vertical) axis should read “numbers of molecules”. Emin is the energy level required for a molecule to jump from the liquid to vapor state. Obviously more molecules have this greater kinetic energy on the higher temperature curve..
If we warmed the water and air, the whole curve shifts to the right and if we cooled it this curve shifts to the left, as indicated.
[As an aside, if you read my essay on “Water“, you will recall that water is a special molecule with a negatively charged side and positively charged side. The negative side of one water molecule is attracted to the positive side of another water molecule in what is called a hydrogen bond. This kind of bond is so important that it would be worth reviewing in that post.]
Let’s begin and see what starts to happen. The most energetic of the molecules in the water have enough energy to break the hydrogen bonds to their neighbors and “jump” up into the air. These are the molecules indicated in the shaded area to the right of the Emin in the above diagram. Since the air is starting out dry, all the movement of the water will be in one direction: from the liquid water below into the air above. It’s evaporating. Once in the air, however, these “new” water vapor molecules will start colliding with air molecules and with each other, and in so doing will gain or lose some of their kinetic energy. Those that lose energy in these collisions may no longer have enough energy to resist the attraction of the negative or positive charge in the water below and may rejoin the liquid water and form hydrogen bonds again. They will be condensing. This exchange happens for awhile, and as it proceeds, the number of water vapor molecules moving back into the liquid water starts to increase because there are simply more of them. Soon, the number of water molecule moving from the liquid up into the air is equal to those moving from the air back to the liquid. The air is saturated. To us, it appears that evaporation stops, though it is really just a new balance in the movement of water between states. The evaporation rate is equal to the condensation rate. If we were to warm the jar with our water and air, there would be more liquid molecules able to jump up in the air and fewer vapor molecules reconnecting with the water. Instead of saying that the air is “holding more water,” it is more enlightening to realize that a new equilibrium is being established with more molecules in vapor form.
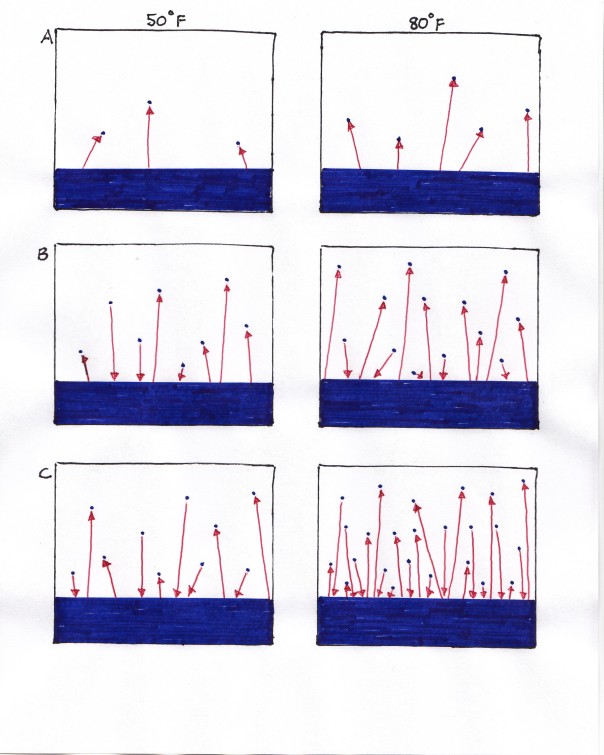
At the lower temperature on the left, equilibrium between evaporation and saturation is established with fewer water vapor molecules in the air than at a higher temperature, shown on the right.
Inside the closed jar, with the pressure steady and the temperature stable (unless we decided to change it), the air eventually became saturated. But in the real world this balance of water molecules going from liquid to vapor and vapor to liquid cannot be established so easily. There may not be a swamp or pond or lake or ocean below the air to act as a water source. The temperature of the water or “parcel” of air may change. We take the top off the jar, drier air blows in and, once again, more water molecules will be jumping up into the new, drier, air.
Ways to Measure the Amount of Water in the Air
So, how does one define how much water is in the air? What ways will be most useful?
Relative Humidity
“Relative Humidity” is the term we are most used to hearing on the weather report, so let’s start there. The amount of water vapor in the air compared to the air itself is not all that great, especially when you compare how differently very humid air feels compared to dry air. It is usually somewhere between 2 and 6 grams of water to one kilogram of air (~4 gm water/1000 gm air). It the warm tropics this number can approach 20 gms of water to one kilogram of air. This measurement is called the mixing ratio. In our ideal example with the jar, we saw that the air became saturated and that the point at which this happened depended only on the temperature and the need for a water source nearby. The ratio of the mass of water vapor at saturation to the mass of the air is the saturation mixing ratio (duh!). In the real world, the actual mixing ratio might be well below the saturation mixing ratio. As I noted above, there might not be a good water source nearby and our air parcel might be blowing all around over land or up in the sky. In fact, the air even over the ocean is rarely at its saturation point. Relative humidity is an attempt to describe how close to the saturation point the air is at. The way this is done is by determining the ratio of the the mass of water in the actual air to the mass it would potentially have at saturation (x 100 to give a percentage).
Relative Humidity = (actual mass of water in a parcel of air/potential mass of water in the air) x (100)
You can imagine that if the relative humidity is low, the potential evaporation rate is high. This is useful in telling us how quickly our clothes will dry or whether our sweat will evaporate.
Dew Point
Dew point is another useful measure of water in the atmosphere. Instead of telling us how close to saturation the air is at a particular temperature, it tells us the temperature at which the air would become saturated if you didn’t change the amount of water vapor in the air.
Dew Point: The temperature at which the condensation and evaporation rate of water vapor are equal and the air is saturated.
Remember the condensation rate increases relative the the evaporation rate as the temperature cools. If I tell you the dewpoint is 50 degrees, I am saying that, given the amount of water in the air, dew will start to form on the grass at 50 degrees. If the temperature does not fall below 50° F, no dew will form. You can see that dew point is an indication of how much water is in the air, but not how much evaporation exceeds saturation. The actual air temperature is usually above the dew point. If the dew point were higher than the actual temperature, the condensation would exceed evaporation, the water vapor would exit the air and form a cloud or form drops on some other surface. Since this is the only real measure we have of the actual amount of water in the air, it is very useful to predict how much rain could potentially fall. An air mass with a dew point of 65°F contains fair more water vapor than an air mass at 45°F. This is something you can now understand when floods are predicted and you see that, indeed, the dew point is high.
How to Make a Raindrop.
There is one thing I failed to mention in the first part of this post which I must now bring to light. I said that the water in the jar will become saturated when the number of water molecules moving from the water to the air is equal to the number moving from the air to the water. But, this is saturation with respect to a flat surface of water. The temperature at which evaporation and condensation rates are the same are different with respect to a curved surface of water!
Huh? Where would one ever encounter a curved surface of water, why would it matter, and would it matter much? Well, I obviously already gave it away in the title to this section. 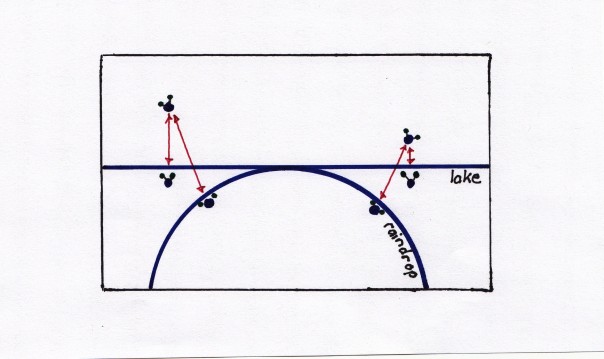 A raindrop is where you would find such a curved surface of water, and to understand how raindrops begin to be formed in the first place, this difference is important. We are going to go back to our old friend, the hydrogen bond, and for this I have drawn a visual aid shown above. In this diagram I have drawn both a flat surface of water (jar, lake, ocean) and a curved surface of water (a raindrop) with two water vapor molecules jiggling over them. This time, however, I have drawn imaginary links (in red) between the charged halves of these molecules to the charged halves of the molecules in the liquid water. These are the electromagnetic forces trying to draw the water vapor back to the surface to reform a hydrogen bond. With the raindrop notice that the liquid water surface curves away from our water vapor molecule, and hence, the forces wanting to pull it into the raindrop are farther away. The strength this electromagnetic force decreases with distance (by the square of the distance to be exact). So, the smaller the water droplet, the harder it is for it to pull in more vapor and grow. This means that the dew point with respect to a curved surface of water is lower than with respect to a flat surface. In fact, as the volume of the droplet shrinks to near nothing, it’s ability to gather more water vapor molecules also shrinks to almost nothing. Even when the air is far below the dew point with respect to a flat surface of water, water will not condense into the raindrop, but will stay as vapor. In this state of affairs, we say the air is supersaturated.
A raindrop is where you would find such a curved surface of water, and to understand how raindrops begin to be formed in the first place, this difference is important. We are going to go back to our old friend, the hydrogen bond, and for this I have drawn a visual aid shown above. In this diagram I have drawn both a flat surface of water (jar, lake, ocean) and a curved surface of water (a raindrop) with two water vapor molecules jiggling over them. This time, however, I have drawn imaginary links (in red) between the charged halves of these molecules to the charged halves of the molecules in the liquid water. These are the electromagnetic forces trying to draw the water vapor back to the surface to reform a hydrogen bond. With the raindrop notice that the liquid water surface curves away from our water vapor molecule, and hence, the forces wanting to pull it into the raindrop are farther away. The strength this electromagnetic force decreases with distance (by the square of the distance to be exact). So, the smaller the water droplet, the harder it is for it to pull in more vapor and grow. This means that the dew point with respect to a curved surface of water is lower than with respect to a flat surface. In fact, as the volume of the droplet shrinks to near nothing, it’s ability to gather more water vapor molecules also shrinks to almost nothing. Even when the air is far below the dew point with respect to a flat surface of water, water will not condense into the raindrop, but will stay as vapor. In this state of affairs, we say the air is supersaturated.
So how does a water droplet get started? This takes something called a condensation nuclei which is nothing more than a fancy name for a dust mote. Almost anything can serve as such a dust mote: soot from a chimney or forest fire, dead phytoplankton from the ocean, clay from a dust storm, sulfates from volcanic activity, perhaps even Whoville. The condensation nuclei acts as a proxy for a flatter surface. This is especially true if this nuclei has a bit of a charge itself and water molecules find themselves attracted to it. As the tiny drop grows larger around this nuclei, it’s surface becomes flatter with respect to the water vapor molecules and condensation happens more easily (the extremely low dew point with respect to a curved surface approaches the more environmentally likely dewpoint of the flat surface).
In the air this usually happens when air cools. For reasons you can’t be expected to know from this post, rising air cools. And soon as this cooling, rising air reaches its dew point temperature, fewer water vapor molecules have the necessary kinetic energy to remain as vapor and will condense onto the nearest surface of water. This is how a raindrop is formed. You may have noticed how condensation forms on the outside of your cold drink on a hot humid summer day. The surface temperature of the glass is far below the dewpoint temperature of the air. Shake some of the drops on the glass off onto your face. You just made it rain.
Clearing the Fog off your Windshield
So let’s see if you can use what you learned here to help you in a real life scenario. You live in Seattle and it is a rainy January evening and the temperature is 45F (6C). You have the windshield wipers on but that’s not helping much because the windows are fogging up from the inside. So, you turn on the heat and aim it toward the windows and circulate the air just within the car to try and dry the windows off (surely, the air in the car is drier that the soggy air outside!). But strangely, that just makes it worse!
What’s going on?
Now you are wiping the windows with your hand to be able to see outside. You stop at your friend’s house to pick them up and a strange thing happens. When you open the door and they get in, a cold damp gust of wind blows in and the windows clear a bit. You open the window a crack. Now you are going to experiment like a scientist. What if you change the source of the heated air blowing on the windows to outside air instead of recirculating the warmer inside air? All this is working wonders. But it’s all very counter intuitive. Blowing cold damp air at the window clears them up? If you want to keep thinking through this problem, don’t read below.
What you have done is to confuse relative humidity with dew point. It seems drier in the car because the relative humidity is lower, while outside the car the relative humidity is nearly 100% and there are even foggy patches. Outside the car the air is nearly saturated with water vapor, inside the car it is not. On the outside, the evaporation rate equals the condensation rate, but on the inside the evaporation rate is greater than the condensation rate. Indeed, judging by relative humidity, the windows should clear up with the windows closed. But at 45F, the air outside has a much lower dewpoint. Since it is foggy out you can see you are nearly there at 45F. Much of the water vapor in the outside air is already condensed out. Inside the warmer car the dewpoint is higher, perhaps 65F. There are many more water vapor molecules in a given volume of air on the inside compared to the outside. The mixing ratio is higher.
So, why does the relative humidity matter less than the dew point? Because the glass on the windows is much cooler than the other surfaces on the inside of the car. It is chilled from outside. The water vapor molecules do not condense on the seats or on your hair. They condense on an object less that the 65F dew point. Bring in some of that lower dew point air in from outside!
Why Do We Sweat?
In my essay on Water I explained that breaking a hydrogen bond, and going from liquid to vapor requires an extra bit of heat energy called the heat of vaporization. This heat energy is sucked away from the molecules in the surrounding environment, which tends to cool the environment. If this evaporation is water on your skin, your skin will cool. Recall that evaporation will happen faster when the relative humidity is low. In a desert, with very dry air, your sweat will evaporate rapidly, helping to cool you off. In a tropical setting with high relative humidity and a low evaporation rate, your sweat just sits there doing nothing. It can’t cool you if it can’t evaporate. This is why 95F in the desert feels much less oppressive than 95F on a muggy day. This same heat of vaporization is released back into the environment during condensation. This can be significant where condensation is occurring quickly like inside a thunderstorm. Stay tuned.
Last Bit of Trivia
At the beginning of this post I posed a question under the photograph of a cloud: Is the cloud composed of water vapor or liquid water? I hope you will now guess correctly that the cloud composed of tiny drops of floating liquid water. Water vapor is completely invisible to the human eye. But there was another plausible guess. What about floating ice? There is an easy what to tell if a cloud is composed of floating drops of liquid water or floating specks of ice. Clouds composed of liquid water droplets tend to have clear boundaries. The drops evaporate rather quickly in the unsaturated air outside the cloud. But ice, with very much less kinetic energy, does not evaporate so easily. They tend to live longer outside the cloud and they give the cloud a “wispy” look.
Posted on May 19, 2013, in Uncategorized and tagged condensation, defog windshield, dew point, evaporation, mixing ratio, raindrop formation, relative humidity, respect to a flat surface of water, sweat, water and ice in clouds. Bookmark the permalink. 5 Comments.



Your explanation of the fogging car windows is a wonderful example of the explanatory power of science. Understand a few basic facts thoroughly, and all kinds of complex situations can suddenly be explained!
love the way you worked Seattle into this. One question? When all the atoms and molecules are ” jiggling” and the most energetic molecules jump, do they do ” Jazz Hands” too? 🙂
Sorry, you know I’m a science dummy.
Crys, I personally think the hydrogen atoms on the water molecule look like Mickey Mouse ears, but I respect your “jazz hands” interpretation #scienceandpoetryinlove
Craig…don’t those tiny drops of floating liquid water have some water vapor in them? As you might have guessed, I chose the wrong answer. You amaze me with all the knowledge you can keep in your head. Unfortunately many things I learned from your article have already left my brain!! Beth
Pingback: Heat Transfer | Craig's Sense of Wonder